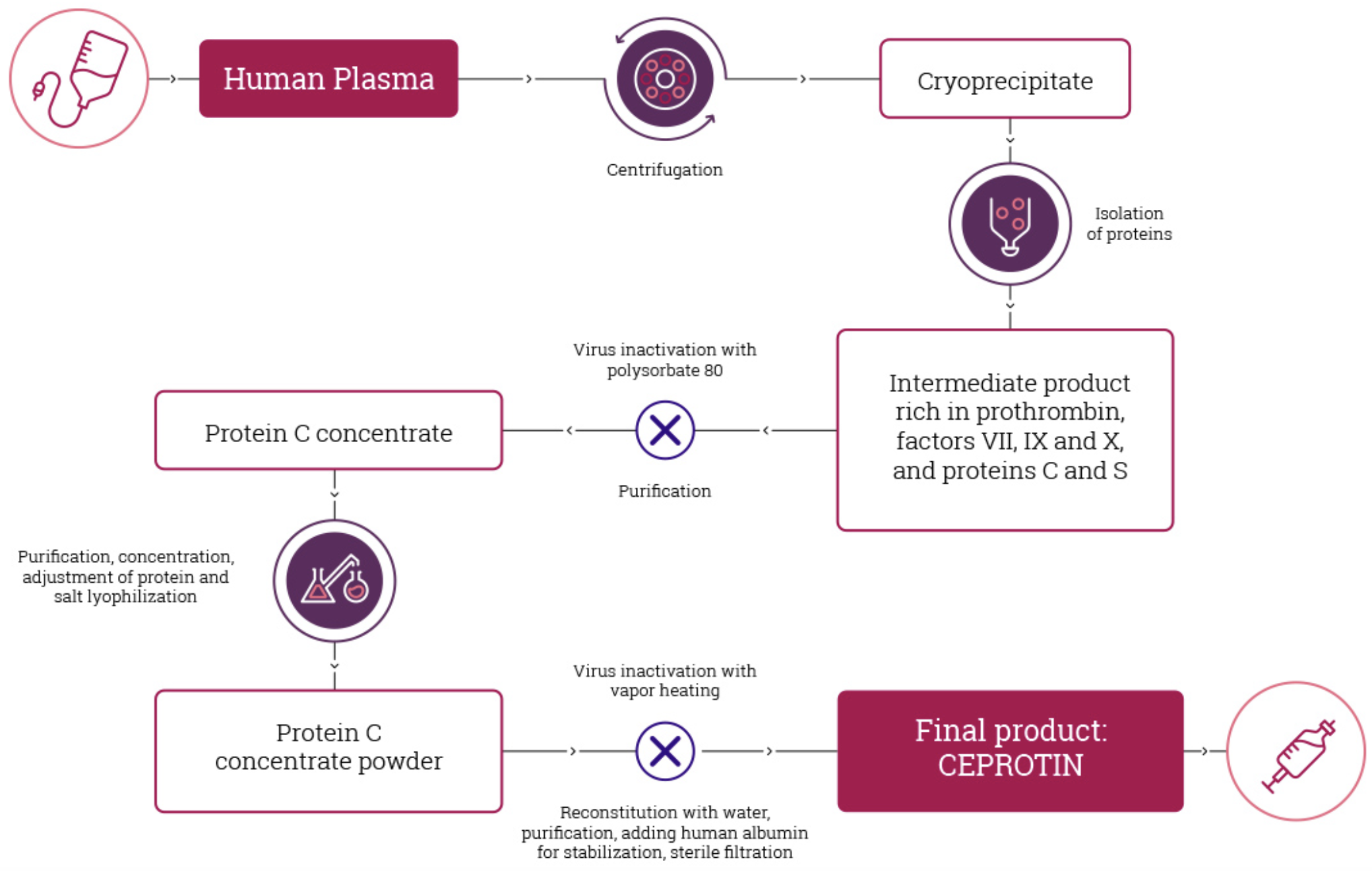
CEPROTIN manufacturing
CEPROTIN is a non-activated protein C concentrate purified from human plasma1,2
CEPROTIN is derived from human plasma and passes through a rigorous manufacturing process to reduce the risk of viral transmission that is normally associated with plasma based products1. CEPROTIN goes through a detergent treatment, heat inactivation and human immunoaffinity chromatography to ensure either the removal or inactivation of human immunodeficiency virus (HIV), hepatitis A, B and C virus and parvovirus B191. Because CEPROTIN is made from human plasma, it may carry a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeld-Jakob disease (vCJD) agent, and theoretically, the Creutzfeld-Jakob disease agent. ALL infections suspected by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Takeda Inc., at 800-828-2088. Discuss the risks and benefits of this product with your patient.1
CEPROTIN manufacturing process3
Warnings and Precautions
Hypersensivity: CEPROTIN may contain trace amounts of mouse protein and/or heparin as a result of the manufacturing process. Allergic reactions to mouse protein and/or heparin cannot be ruled out. If symptoms of hypersensitivity/allergic reaction occur, discontinue the injection/infusion. In case of anaphylactic shock, the current medical standards for treatment are to be observed.
Transmission of infectious agents: Because CEPROTIN is made from human plasma, it may carry a risk of transmitting infectious agents, e.g. viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and theoretically, the Creutzfeld-Jakob disease (CJD) agent.
Bleeding episodes: Several bleeding episodes have been observed in clinical studies. Concurrent anticoagulant medication may have been responsible for these bleeding episodes. However, it cannot be completely ruled out that the administration of CEPROTIN further contributed to these bleeding events. Simultaneous administration of CEPROTIN and tissue plasminogen activator (tPA) may further increase the risk of bleeding from tPA.
Heparin-induced thrombocytopenia (HIT): CEPROTIN contains trace amounts of heparin which may lead to HIT, which can be associated with a rapid decrease of the number of thombocytes. If HIT is suspected, determine the platelet count immediately and consider discontinuation of CEPROTIN.
Low sodium diet/Renal impairment: Patients on a low sodium diet or who have renal impairment should be informed that the quantity of sodium in the maximum daily dose of CEPROTIN exceeds 200 mg. Monitor patients with renal impairment closely for sodium overload.
Adverse Reactions
Common adverse reactions related to CEPROTIN observed in clinical trials were hypersensitivity or allergic reactions: lightheadedness, itching and rash.
INDICATION
CEPROTIN [Protein C Concentrate (Human)] is indicated for neonates, pediatric and adult patients with severe congenital Protein C deficiency for the prevention and treatment of venous thrombosis and purpura fulminans.
Please click for Full Prescribing Information.
References:
-
CEPROTIN [Protein C Concentrate (Human)] Prescribing information. Lexington, MA: Baxalta US Inc.
-
Goldenberg N, Manco-Johnson M. Protein C deficiency. Haemophilia. 2008;14(6):1214–1221.
-
Knoebl PN. Human protein C concentrates for replacement therapy in congenital and acquired protein c deficiency. Drugs Today. 2008;44:429-441.